|
3/19/2023 0 Comments Atomic radius of calciumThe temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. 
The arrangements of electrons above the last (closed shell) noble gas. 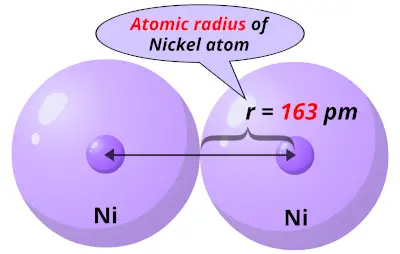
These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. If anything, the radius of the atom, along with electron repulsion and effective nuclear charge determines an atoms's electronegativity (for example, higher nuclear charge and small radius will want electrons while a larger radius has less of a tight hold on it's valence electrons and will most likely give those valence electrons away).A vertical column in the periodic table. Electronegativity does not play a role in creating the ionic radius or atomic radius. I don't think atomic weight affects the radius due to the the weight being caused by the amount of protons and neutrons. Overall ionic radius show the same trends as the atomic radius (increase down a group and decrease across a period) but there may be more components to take into account for non-isoelectronic atoms when comparing their radii. The Ca2+ ion has valence electron in the 3rd shell while is further from the nucleus, therefore making its ionic radius larger than Sodium's. Although it loses two electrons, Calcium will have an electron configuration of which ends with 3p6 while Na+ has the electron configuration of which ends with 2p6. However, if you compare their sizes Calcium will have a larger ionic radius because it is had more shells to begin with and more electrons compared to Na. By losing electrons, there is a loss of extra electron repulsion and the effective nuclear increases for both which will pull the electrons in closer to the nucleus. With your example of Na+ and Ca2+, both atoms lose electrons to form cations. Nuclear charge, the number of shells, shielding and electron repulsion I think are what contribute the most to atomic radius. As you move down a group, additional shells are added to the atoms electron configuration which are further from the nucleus, so they make the atoms larger, and the electrons are less tightly held in their valence shell.Ītomic radius also decreases across a period because the nuclear charge increases across a period which pulls the electrons more tightly since they all have electrons in the same valence shell. The general trend for atomic radius is that it increases down a group and decreases across a period. Rather the atomic radius determines the electronegativity. However, if an atom gains an electron from its high electronegativity, electronegativity does not play a role in deciding the ionic radius. Electronegativity is how much an atom wants an electron. I don't believe the larger atomic weight plays a role in atomic radius, but I could be wrong. I'm not exactly sure if we would have to compare the atomic radius of ions that are non-isoelectronic because I believe there are more complexities, but the ionic radius show the same overall trends as the atomic radius. Moreover, the calcium ion still has more shells which means it is further from the nucleus, so the calcium ion would have a larger ionic radius. I believe that the calcium ion has a larger ionic radius, and this is my thought process:Īlthough the calcium ion lost 2 electrons, it still has more electrons than the sodium ion so there is still some electron-electron repulsion compared to the sodium ion. This is why cations become smaller than their parent atoms. When both atoms loses electrons, this leads to less electron-electron repulsion, so it pulls electrons more tightly. The sodium ion has 10 electrons and 11 protons. When 2 electrons are removed the Calcium ion has 18 electrons and 20 protons. With your example of Na+ and Ca2+, you can look and see how Calcium has 20 protons and Sodium has 11 protons. I think effective nuclear charge, the number of shells, and electron repulsion are very important when discussing atomic/ionic radius. This is because the nuclear charge increases (there are more protons) which pulls the electrons more tightly. 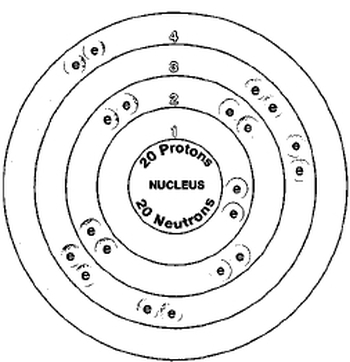
These additional shells are further from the nucleus, so they make the atoms larger, therefore their atomic radius is larger.Ītomic radius also decreases across a period. Atomic radius ultimately increases down a group because there are additional shells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
